Field Asymmetric Ion Mobility Spectrometer (FAIMS) provides rapid (30 seconds for a sample) analysis under atmospheric pressure.
* corresponding author (chantipmanee.n@hotmail.com)
Received: 28 February 2020, Accepted: 06 June 2020, Published: 31 December 2020
DOI: 10.5281/zenodo.4383331 | 📁 view PDF file
Background
Field Asymmetric Ion Mobility Spectrometry (FAIMS) is one of the Ion Mobility Spectrometry (IMS) techniques. FAIMS is a very sensitive technique that can identify gaseous phase ions under atmospheric pressure. FAIMS has been developed for decades for simple and fast analysis applied to a broad range of applications in military, agriculture, and pharmaceutical research (Eiceman, Karpas, and Hill 2013, Kolakowski and Mester 2007). Most of the compounds of interest are Volatile Organic Compounds (VOCs) (Chavarria et al. 2017). Separation and identification of VOCs at atmospheric pressure can be achieved. In the work, benzene and toluene as VOCs were identified by FAIMS. Each VOC is transported in the air as carrier gas by a UV lamp with a specific wavelength to ionize analytes of interest. The ions were passed through a separation region which consists of two parallel electrodes. One electrode was connected to an asymmetric waveform. The trajectories of ions were dispersed by this oscillating waveform. Only the ions with specific mobility can move through and reach the ion detector.
Method
Analytes of interest - Benzene 5 µL·L-1 and Toluene 100 µL·L-1 (Calibration gas, Peterson Environmental, Inc, USA) were used as standard gas. Each analyte was introduced individually into the system from a gas tank and the flow rate was regulated by using a mass flow controller (MFC) (F-201CV-1K0-AAD-22V, Bronkhorst High-Tech BV, Netherlands). The analyte was merged with 9 L·min-1 air as dilution and carrier gas to obtain an exact concentration of 200 nL·L-1. 500 Vpeak-to-peak and 800 kHz as asymmetric waveform was connected to the top electrode and voltage scanning from -15 to +15 V as compensation voltage was attached to the bottom electrode as shown in the inset A of the Figure. Peak profiles were plotted as the FAIMS signal against the compensation voltage.
Result
As shown in insets B and C of the Figure, the analytes were ionized by a UV lamp (Heraeus PKS106, Heraeus Korea Corp, Germany) with radiation energy of 10.6 eV. The gaseous analytes were moved to the separation region made of two pieces of the printed circuit board (PCB) separated with a spacer of 0.3 mm to provide an ion channel as shown in inset A of the Figure. The top electrode was connected to an asymmetric wave to oscillate the ions, whereas the bottom electrode was connected to compensation voltage scanned from -15 to +15 V to compensate the waveform allowing specific ions going through, and reach the detector. The signal was recorded with an amplifier with a gain of 9×109 V/A. Peak position as shown in inset B and C of Figure was observed and allowed the identification of each ion according to the FAIMS principle. The position of compensation voltage identified which kind of VOCs is present. Peak profiles of benzene (Molar mass: 78.11 g/mol) at 4.62 V with equation of y =(0.1111 ± 0.0017)+ (0.0314 ± 0.0315); r2= 0.9998 and toluene (Molar mass: 92.14 g/mol) at 4.51 V with equation of y =(0.1559 ± 0.0005)+ (0.1046 ± 0.0708); r2= 0.9989 were obtained.
Discussion and conclusion
The performance of FAIMS for selection of benzene and toluene has been successful. A low concentration down to 200 nL·L-1 could be detected. The system provides automatic operation in 30 seconds for a cycle. The FAIMS is a promising technique to be developed as a miniaturized device.
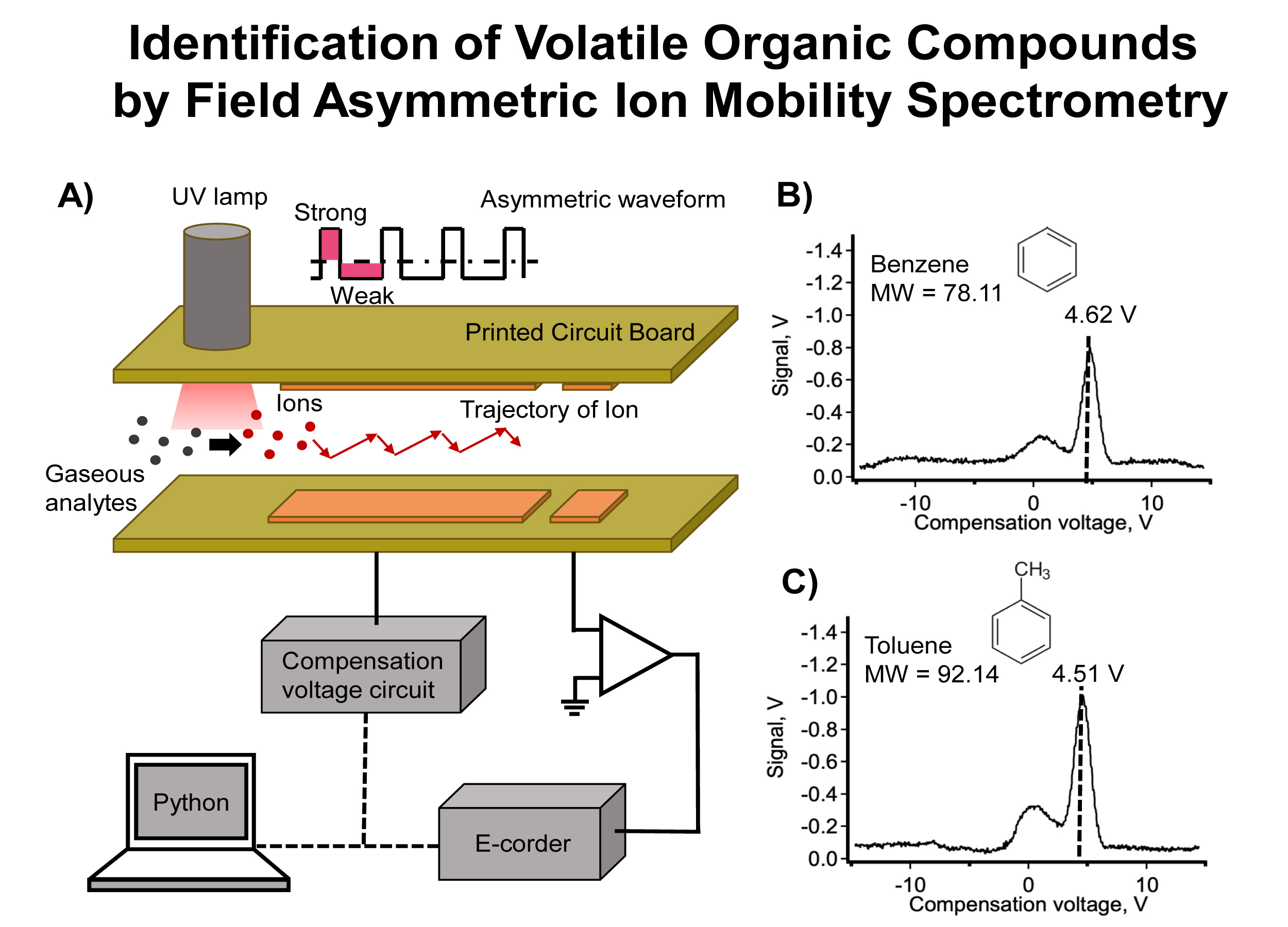
A) Schematic diagram of VOCs movement in Field Asymmetric Ion Mobility Spectrometer and its connection to obtain peak profiles of B) benzene (MW = 78.11) at 4.62 V and C) Toluene (MW = 92.14) at 4.51 V
About the authors
Mr Nattapong Chantipmanee is a doctoral student under the supervision of Prof. Peter C. Hauser at the Department of Chemistry, Basel University, Switzerland. His current research area has been the development of electrospray ionization mass spectrometry (ESI-MS) and of ion mobility spectrometry (IMS) for inorganic ions.
Acknowledgements
NC would like to acknowledge the support by the Development and Promotion of Science and Technology Talents Project (Royal Government of Thailand scholarship) (DPST: 532133) and by Association of Thai Students in Switzerland (ATSS).
References
- Chavarria, M. A., A. V. Matheoud, P. Marmillod, Y. Liu, D. Kong, J. Brugger, and G. Boero. 2017. “High sensitivity field asymmetric ion mobility spectrometer.” Review of Scientific Instruments 88 (3). doi: 10.1063/1.4978960.
- Eiceman, G. A., Z. Karpas, and H. H. Hill, Jr. 2013. Ion mobility spectrometry, third edition, Ion Mobility Spectrometry, Third Edition. Book.
- Kolakowski, B. M., and Z. Mester. 2007. “Review of applications of high-field asymmetric waveform ion mobility spectrometry (FAIMS) and differential mobility spectrometry (DMS).” Analyst 132 (9):842-864. doi: 10.1039/b706039d.
License
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0) License, which permits to copy and redistribute the material in any medium or format. You are also allowed to remix, transform, and build upon the material under the following terms: 1) You must give appropriate credit, provide a link to the license, and indicate if changes were made. 2) You may not use the material for commercial purposes. 3) If you remix, transform or build upon the material, you must distribute your contributions under the same license as the original. To view a copy of this license, visit https://creativecommons.org/licenses/by-nc-sa/4.0/